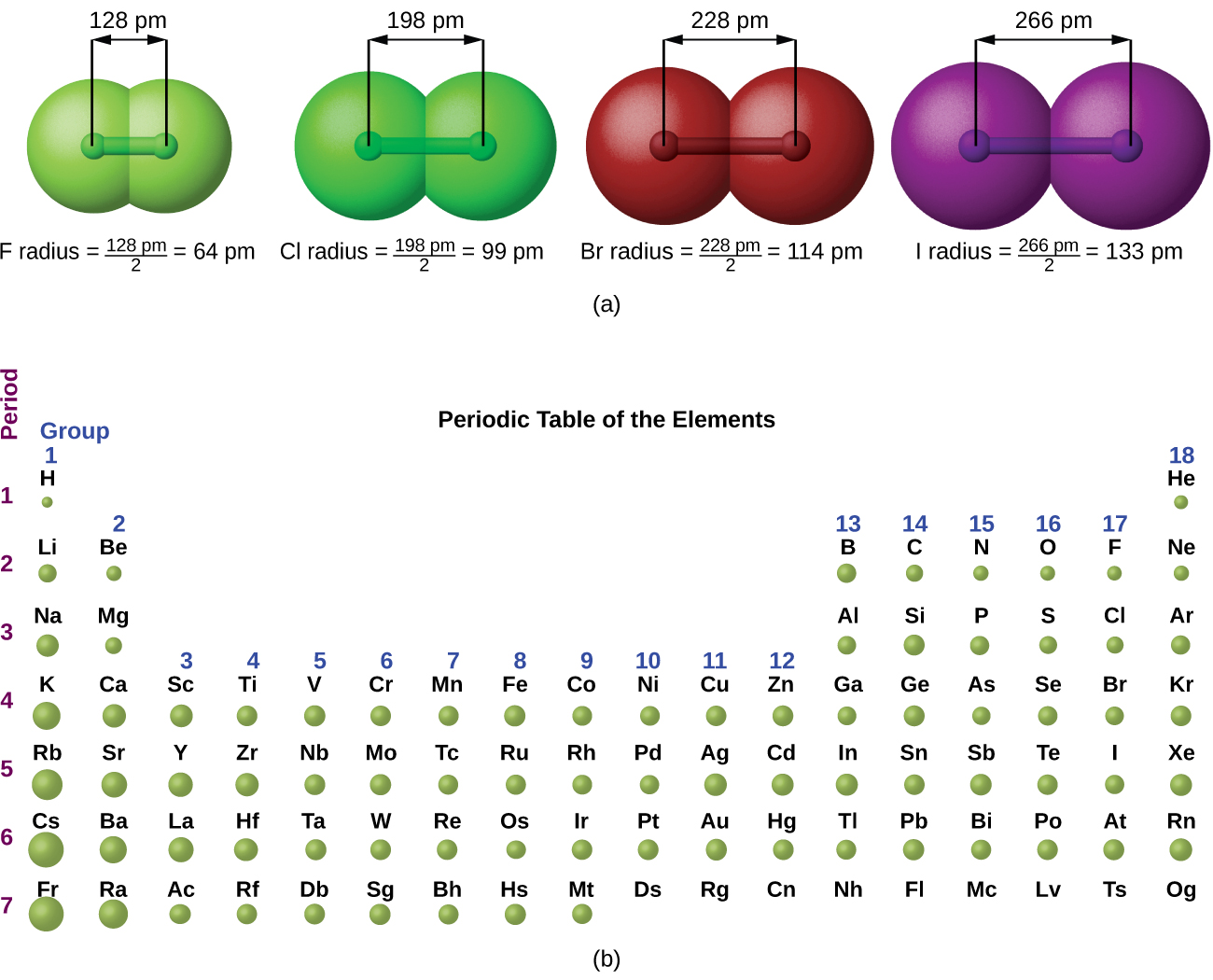
Sodium is the most abundant alkali metal and makes up about 2.6 percent by weight of the Earth's crust. In medieval Europe, a compound of sodium with the Latin name sodanum was used as a headache remedy. That name in turn comes from the Greek nítron, a kind of natural salt. The symbol for sodium, Na, comes from the neo-Latin name for a common sodium compound called natrium. 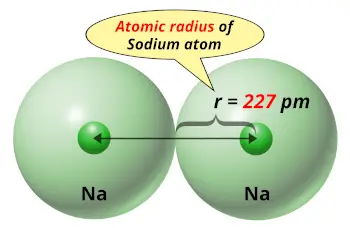
The name sodium comes from the English word, soda. The metal itself was first isolated in 1807, when Sir Humphry Davy carried out the electrolysis of caustic soda (sodium hydroxide). The carbonate, chloride, and hydroxide of sodium are important industrial chemicals, used in the manufacture of glass, pulp and paper, textiles, soaps and detergents, and other sodium salts.Ĭompounds of sodium have long been known. Sodium hydroxide is a powerful chemical base, but sodium bicarbonate and sodium carbonate are also used to raise the alkalinity of solutions. Sodium bicarbonate is an antacid, a leavening agent in baking, and an ingredient in some forms of toothpaste. It is also an agent for de-icing highways in the winter. It provides the body with sodium ions, which are essential nutrients, but excessive amounts are harmful for health. Common salt, or table salt, is sodium chloride, widely used as a food flavoring. Moreover, the compounds of sodium have numerous applications. An alloy of sodium and potassium (NaK) is a heat-transfer medium and a chemical reducing agent. Sodium vapor lamps provide bright illumination at low cost. Sodium metal can be used to isolate some other metals from their compounds, smoothen metal surfaces, and manufacture several compounds. Given its reactivity, sodium is not found as a free metal in nature but is bound to other elements in the form of compounds. It is highly reactive, oxidizing rapidly in air and reacting violently with water to produce the alkali sodium hydroxide and hydrogen gas. Silvery in color, it is soft, waxy, and lightweight. Sodium (chemical symbol Na, atomic number 11) is a member of a group of chemical elements known as alkali metals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
